Courtesy of highlands.edu
The diagnosis of a neurological disease can represent what many patients see as a point of no return, but research published on Thursday, Oct. 25 by UR scientists on stem cell therapy points toward a potential for lessening the severity of the outcomes of these diagnoses.
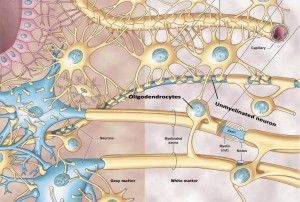
The research, which was published in the journal “Science,” was conducted by UR researchers Steve Goldman, Maiken Nedergaard and Martha Windrem, addresses stem cells capable of transforming into the brain’s two main support cells, astrocytes and oligodendrocytes. The latter is lost in a family of neurological diseases classified as myelin disorders.
Myelin disorders include Multiple Sclerosis, Cerebral Palsy and Tay-Sachs disease, among others. These diseases are well known for being life-altering, so research with implications for dampening their effects will have an important impact on patients and their families once brought to the practical level.
This particular type of cell therapy would rely on programming stem cells to propagate glial progenitor cells. Progenitor cells then give rise to oligodendrocytes, which insulate neurons with a fatty substance called myelin. In total, this process ensures the speed and efficiency of neuronal communication. Without myelin, nerve communication throughout the body becomes weakened, which is the underlying pathology that causes the slowed cognitive processes and muscle dysfunction that are characteristic of myelin disorders.
Goldman’s lab was instrumental in understanding the chemical signals necessary to not only turn stem cells into progenitor cells, but also to ensure that they specifically produce oligodendrocytes.
According to Goldman, the team’s research focused on using stem cells to create glial progenitor cells purposely because they are less variable than other cells found in the central nervous system and thus present a relatively direct target.
This research on myelin replacement, although pointed toward a specific goal in the end, actually derives from research of something seemingly unrelated: song birds.
“We worked primarily on stem cells of the brain that give rise to new neurons to try and understand how they progenerate regions of damaged brain, and that’s work that started and still continues in song birds,” Goldman said. “Every time they learn a new song, their brains lay down a new bunch of neurons that control the song. There’s been a lot of effort over the years to understanding the biology of how birds sing, which then led to studies of brain progenitor cells, not only in birds, but in mammals, and eventually humans. That’s when we found out about brain progenitor cells that give rise to oligodendrocytes and astrocytes instead of neurons.”
Goldman and his colleagues’ research has been taken to the brink of human clinical trials, which is now the final frontier for the practical application of this therapy.
“What’s left for this study is to start clinical trials,” Goldman said. “But that’s not far off. At this point, taking that next step just relies on acquiring funding.”
Sklar is a member of the class of 2014.